It has long been known that a result of unresponsive or persistent pain conditions is sensitization of the central nervous system; which essentially means that the individual experiencing it has both a lower pain threshold and a higher pain perception. For example, someone who has a history of ongoing pain in their elbow may bump their elbow as they walk through a doorframe and perceive it as very painful; whereas someone without that same pain history may barely notice that they bumped their arm at all. This is an example of lower pain threshold.
Similarly, if these two people were to fall on their elbow on concrete (a typically painful event), the first sensitized individual may perceive it as extremely painful and longer term and the second non-sensitized individual as moderately painful and shorter term. This is an example of increased pain perception based on the same level of tissue injury.
More recently, however, this same central sensitization has been identified in people with other persistent health challenges, such as irritable bowel syndrome, tension headaches, premenstrual syndrome, temporomandibular joint (TMJ) dysfunction (also known as TMD) or fibromyalgia – amongst others, see below.
Sometimes termed, central sensitivity – this sensitisation has now been recognised to occur not only in those with pain, but also in those with lower level – but persisten – symptoms, such as bloating, pruritus (itchy skin), brain fog or tinnitus (ringing ears), anxiety, therefore being a feature of many more conditions and affecting many more people than originally thought.
What is Central Sensitivity?
For some decades now, the term central sensitization has been broadly recognised in the medical field – simply meaning, as the name suggests, a sensitization of the central nervous system. However, the term central sensitivity was first coined by Rheumatologist, Muhammed Yunus MD in 2008, and was used to denote the common link between many different chronic widespread pain syndromes often used as “place-holder” terms for patients whose conditions seem to evade a clear diagnosis. Historically, people experiencing these strange symptoms and pain may have been given diagnoses, such as:
- functional syndromes
- functional somatic syndromes
- medically unexplained symptoms
- somatoform disorders
- somatization disorder
- psychosomatic syndromes
- idiopathic pain disorder
In the field of back pain, there has been a parallel trend toward a very non-descript, unhelpful and overused diagnostic term: “non-specific low back pain“. In essence, non-specific low back pain (sometimes abbreviated to NSLBP) is simply stating that there is no obvious or specific cause one can point to for the pain experience (such as a fracture, or a disc herniation). Instead, there are multiple findings – sometimes conflicting or contradictory – and a plethora of other factors in the surrounding millieu. A diagnosis of “non-specific low back pain” is therefore most likely to arise from someone who simply does not have the tools or medical literacy to be able to unpick, prioritise and address the millieu of contributing factors.
Threshold effect
Instead of there being “no cause”
Stress is an important part of life; as Hans Selye, the man who coined the term “stress” states:
“The complete absence of stress, is death”
So nobody wants to be completely stress-free! Here’s a some reasons why:
What does physiological load do? Well, loads within your adaptive range (ie loads that do not exceed your ability to adapt) will generally stimulate the system positively encouraging growth – a good example of this is the “overload” principle used in resistance training or the “SAID” principle (Specific Adaptation to Imposed Demands).
Physiological Load & Central Sensitivity
In the study of persistent musculoskeletal pain, several factors have been shown to play important contributory roles. These include (but are not limited to) the patient’s:
- beliefs around pain (especially fear-avoidance behaviours) (O’Sullivan 2011)
- disrupted sleep patterns (Jank et al 2017)
- life stressors / emotions (especially anxiety and depression) (McBeth et al 2007)
- gender (females are more affected) (Dodds et al 2016)
- age (sensitivity to pain increases with age) (Kato et al 2017)
- propensity to central sensitization (Yunus 2008)
Central sensitization is often described as increased responsiveness of nociceptive neurons in the central nervous system to their normal or subthreshold afferent input (Loeser & Treede 2008). While there is general agreement that a nociceptive event must occur to trigger central sensitization (with increased responsiveness of nociceptive neurons in the central nervous system to their normal or subthreshold afferent input (Loeser & Treede 2008). While there is general agreement that a nociceptive event must occur to trigger central sensitization (with other neurological drives contributing to perpetuate it), it is also suggested that a summation of multiple lower grade inputs may initiate (Yunus 2007). These inputs can include peripheral drives (of multiple or singular, visceral or somatic origin), and/or descending drives from suprasegmental influences (Urban & Gebhart 1999, Monaco et al 2017) including limbic-emotional contributions (Yunus 2008).
As such, adverse stress, or what may be termed “physiological load” on any of these systems (visceral, somatic, suprasegmental) can summate reducing nociceptive thresholds – meaning pain can be perceived at lower levels of stimulation, or secondary to non-noxious stimuli, or due to stimulus outside of the damaged area (Latremoliere & Woolf 2009).
Whilst this description of central sensitization’s effects is broadly accepted, its direct effect on motor control is not so often considered. Firstly, the pain – itself a product of central sensitization – will affect motor control, while sensitization within the cord has been shown to affect local postural reflexes (Willard 2001) and is observed clinically to affect muscle firing (Bouhassira 2001, Tremolaterra et al. 2006, Wallden 2005, 2013). Since central sensitization remains below conscious perception (Willard 2001), one role of the clinician may be to bring it to the awareness of the patient with strategies to effectively address it.
As motor control plays an important role both in the function, and in the rehabilitation of the musculoskeletal system (Panjabi 1992, Tsao et al 2011, Maher et al 2017), an unconscious compromise at the cord level may prove a major confounder in research studies focused on motor control in isolation, such as those assessed in the recent systematic review by Maher et al 2017.
CS – Panjabi’s Model from Matt Wallden on Vimeo.
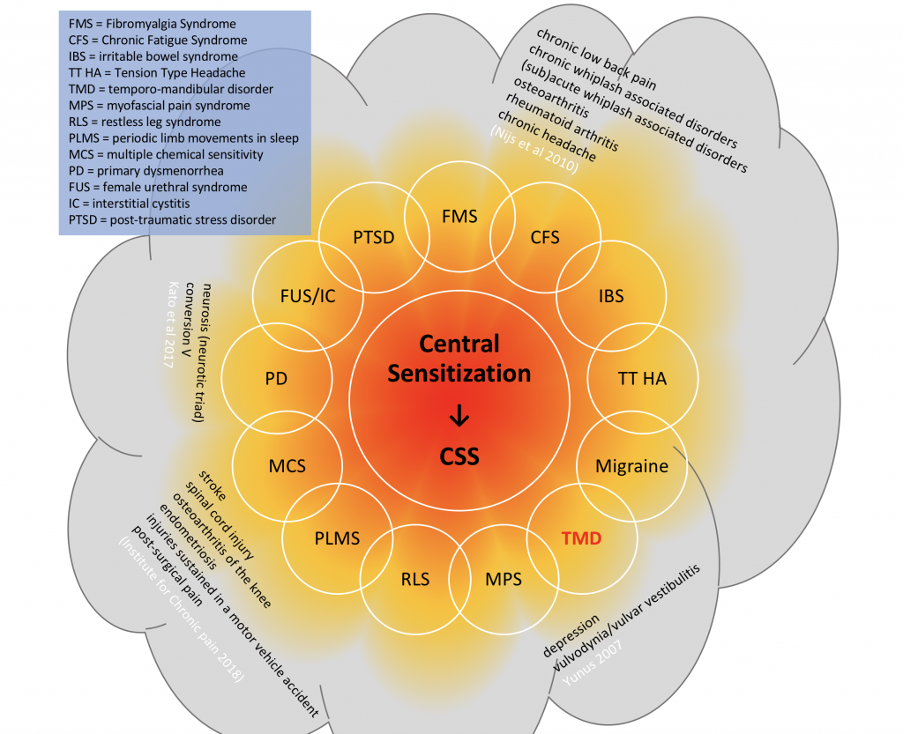
There is a growing list of clinical conditions already cited in the published literature that have been demonstrated to be associated with central sensitivity (Institute of Chronic Pain 2018, Nijs et al 2010, Yunus 2007, 2008), listed in Figure 3b. This points to the role of a holistic integrated approach to working with musculoskeletal patients.
Kindler et al (2011) perhaps rightly urge caution regarding the generalizability of the “central sensitivity syndrome”. Nevertheless, Yunus (2007) observes that central sensitivity (so commonly believed to be the result of pain, trauma or inflammation) may, in fact, manifest even from minor irritation.
Furthermore, Willard (2001) highlights that although sensitization in the dorsal horn arises from nociceptive drives in B-afferent fibers returning from visceral and somatic tissues, A-afferents (with a lower non-nociceptive threshold) can influence local muscle postural reflexes due to some central fibers having direct input into the ventral horn. Hence, low levels of somatic or visceral irritation may adversely affect motor control, while higher, more persistent irritation may drive central sensitization, thereby reducing pain thresholds and compromising function of the motoneuron pool (He et al 1988).
A key clinical point is that although nociceptive firing of the B-afferent fibers is required to initiate sensitization, once sensitization is established, it may be maintained and perpetuated through other sensory stimuli (Willard 2001, Yunus 2007). Associated with this, as highlighted by Moseley (2003), nociception does not equal pain. Nociceptive drives from an inflamed tissue (especially from viscera) do not necessarily reach conscious perception; so symptoms of inflammation, such as bloating or abdominal distension, may be a more clinically useful tool to establish if visceral drives could be contributing to central sensitivity.
Since tonic motoneurons have a lower threshold to stimulus than phasic, it is the tonic fibers in musculature supplied by the affected segment(s) that are most likely to be inhibited by sensitization at the cord level. Lederman (2000), in arguing against the notion of sensitization, suggests it would only occur in a functionally selective way; there would have to be an advantage at the survival level, for which he could see none. However, the benefits would be that if the afferent stimulus were coming from an injured muscle, or from an inflamed joint or disc, then inhibition of tonic activation of the damaged muscle, or of muscles around the inflamed joint/disc would reduce loading and pain – placing any activation under the more conscious control of the phasic system (for further discussion see Wallden 2008). If the afferent stimulus were originating from an inflamed organ, such as occurs in IBS or dysmenorrhea, inhibition of tonic motoneurons would decrease compression of the organ; as seen in the bloating associated with these conditions (for further insights see Wallden 2013).
One of the key findings in the field of motor control, is that pain inhibition occurs to low threshold (tonic) motor units acutely after onset of pain; and that reactivation of these motor units is not automatic, even after the pain has gone (Hides 1996). The pain perpetuation caused by central sensitization has, therefore, both neurological and mechanical ramifications; as motor control will be compromised by ongoing presence of pain. Beyond pain, central sensitization itself may also compromise motor control due to inhibition of motor units as a result of other inputs, such as visceral afferent drives (Bouhassira 2001, Tremolaterra et al. 2006, Wallden 2005, 2013). This sensitization, which remains below conscious perception (Willard 2001), in turn, may result both in provocation or perpetuation of nociception and in altered musculoskeletal pain itself; a broadly recognized process (see Wallden 2013 for more details).
Tennis elbow, Achille’s injury, shoulder pain and symptoms / markers of cardiovascular disease risk point to both central sensitivity and even more-so, to physiological load… What’s the difference between Central Sensitivity & Physiological Load?
A growing body of evidence points to the notion that many musculoskeletal conditions may have their roots in cardiovascular disease risks; specifically dyslipidemia and the atherosclerosis process. In particular, lateral epicondylitis, carpal tunnel syndrome, and shoulder disorders have been identified as having a strong relationship with cardiovascular markers (Hegmann et al 2017). The interesting thing here is that all of these conditions are in the upper limb, which is also affected by cardiac referred pain; hence, central sensitisation from a distressed heart (think angina or a subclinical pre-angina state) may be a driver of the reduced pain tolerance of these upper limb biomechanical conditions, and/or a driver of increased pain perception in the sensory cortex.
Emotions affecting locomotor system (Holstege (2001) and Pert)
Webinar
Assess your physiological load (free / paid-for /
Link to emotional anatomy (anatomy of the spirit).
What’s your physiological load? And why does it matter?
Physiological load is a term which describes the “stressors” on your system.









